PH Domains
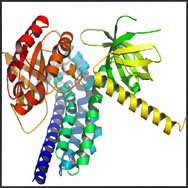
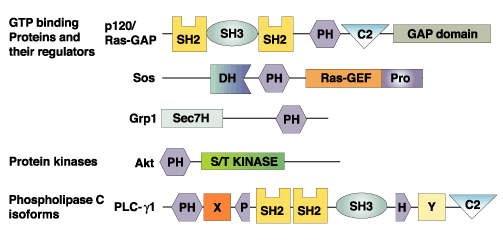
Pleckstrin homology (PH) domains are about 120 amino acid-long protein modules that were first described in pleckstrin, the major protein kinase C substrate in platelets. PH domains have since been identified in several key regulatory proteins with characteristic structural features that include two orthogonal beta sheets that form alpha sandwich with an a helix at the COOH terminus, and variable loops that create a highly charged surface. It has been generally accepted that PH domains provide a structural basis for the interaction of certain regulatory proteins with membranes. Some PH domains bind with high affinity (low µM or nM K d ) to specific phosphoinositides such as phosphatidylinositol- 4,5-bisphosphate, PI-3,4-P2 or PI-3,4,5-P3. Binding to phosphoinositides may allow PH proteins to respond to lipid messengers for example by relocation to membranes. The C-termini of some PH domains have also been reported to bind the beta/gamma subunits of heterotrimeric G-proteins. For example, the PH domain of phospholipase C (PLC) delta binds inositol 1,4,5-tris-phosphate (Ins[1,4,5]-P3 ) and associates with lipid vesicles containing phosphatidylinositol 4,5-bisphosphate (PtdIns-[4,5] P2 ), but only weakly binds other inositol phosphates and PtdIns(4)P. Similarly, the PH domain of the Akt protein kinase appears to bind PtdIns(3,4)-P2 but not Ptd-Ins(4,5)-P2. Because different PH domains specifically bind specific phosphoinositides, PH domains (usually with a fluorescent tag like GFP) have widely used to label the metabolism and distribution of these phosphoinositides.
|
PH Domains |
Phosphoinositides Specifically Labeled |
|
Akt/PKB Ser/Thr Kinase |
PI-(3,4)-P2 |
|
Phospholipase C-delta; mSos1 |
PI-(4,5)-P2 |
|
Btk Tyr Kinase; Grp1 |
PI-(3,4,5)-P3 |
|
Phospholipase C |
Ins[1,4,5]-P3 or PI[4,5]-P2 |
|
|
|
|
|
|
|
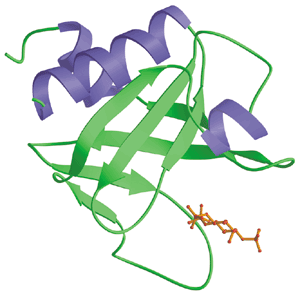
Structure of the PH Domains |
The PH Domain (Green) from Rat PLC delta1 Bound to Ins[1,4,5]-P3 (Red) |
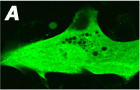
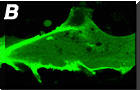
An example showing membrane translocation of GFP tagged PH
domain (Akt) after application of PDGF
|
|