SignaGen Laboratories (SignaGen) is a small business focusing on developing and manufacturing gene delivery tools for bio-medical research community. Based on our unique technology platforms (patent pending, US PTO # 61135606 & 072308), SignaGen has successfully developed three categories of DNA/RNA transfection reagents, which were validated to beat the leading products easily with extremely high efficiency & low cytotoxicity. What’s more important is we are supplying these transfection reagents with logical prices.
1. Biodegradable Polymer (BDP) Synthesis:
This BDP technology is our patent pending (US PTO # 61135606 & 072308) propriety technique which allows us to synthesize a biodegradable polycationic polymer. Unlike other available cationic polymers such as poly(L-lysine) and poly(ethylenimine), and cationic liposome, this novel polymer has an unique backbone which can be degraded after the gene vector unloads DNA/RNA inside of mammalian cells (Figure 1), leading to extremely low cytotoxicity. In addition, this novel polymer was conformed to possess excellent nucleic acid binding affinity and high buffering capacity, meeting the fundamental design criteria of good gene carriers. The in vitro DNA transfection experiments showed that this novel biodegradable polymer was 2 to 10 times more efficient than poly(ethylenimine) in delivering DNA to HEK293T cells.
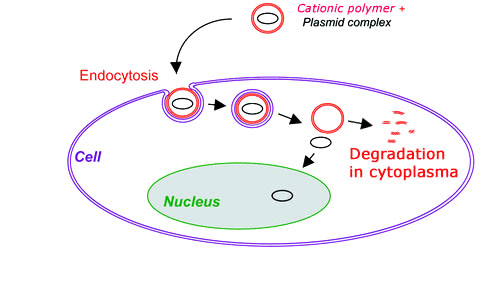
Figure 1. A Cartoon Showing DNA Transfection with A Biodegradable Polymer
2. Virus Proteins Simulating (VIPs) Technology:
Gene therapy is advanced technology in the treatment of human genetic and acquired diseases through the delivery and expression of therapeutic gene-based drugs. The use of safe, efficient, and controllable gene carriers is one prerequisite for the success of clinical gene therapy. While viral vectors are very efficient in gene delivery, their potential safety and immunogenicity concerns raise their risk in clinical applications. As an alternative to viral vectors, we developed an unique VIPs technology to simulate virus nuclear targeting peptide sequences. Based on this VIPs platform, series of simulated virus (e.g., lentivirus) nuclear targeting peptide sequences are created, synthesized and screened. It was confirmed that combined with a liposome DNA transfection reagent, series of these simulated peptides were able to penetrate cell nuclear pore, thus delivering DNA/RNA directly to cell nuclear (Figure 2). The initial results showed that several simulated peptides can significantly (up to 100 times) enhance liposome based gene delivery efficiency on variety of mammalian cells. Interestingly the synergy effect was observed on hard-to-transfect non-dividing cells, providing a potential non-viral nucleic acids vector for hard-to-transfect cells.
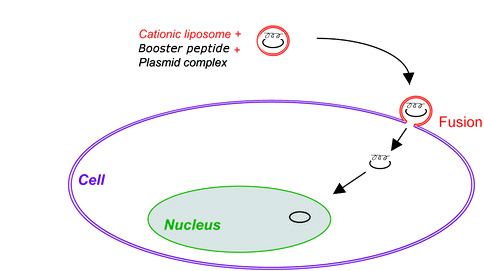
Figure 2. A Cartoon Showing Synergy Effect of VIPs Simulated Peptide with a Liposome Transfection Reagent
3. Shaved Cell Transfection (SCT) Technology:
Cell membrane architecture is that of a lipid bilayer. The lipids are amphipathic in that they have hydrophilic polar heads pointing out and the hydrophobic portion forming the core. However cell membrane surface is uneven (Figure 3-I). Lots of peripheral membrane molecules including glycolipids, glycoproteins, carbohydrate of glycolipids and transmembrane proteins, ion channels are also constituents of membrane. In addition, most cells are decorated with other several types of proteins that allow their binding to other cells or to the extracellular matrix. They are known as cell adhesion molecules (CAMs). All these peripheral membrane molecules are termed as cell’s hairs (Figure 3-I), which hinder transfection complex (Figure 3-II & III) accessing and penetrating the cell, compromising transfection complex endocytosis. This is why regular transfection procedure usually leads to lower transfection efficiency especially on the hard-to-transfect cells. To circumvent barrier of cell’s hair in preventing transfection complex endocytosis, we developed unique transfection reagents with proprietary transfection protocols. With our proprietary transfection procedures called “Shaved Cell Transfection (SCT)”, cell’s hairs are temporarily shaved before conducting transfection followed by re-plating the shaved cells (Figure 3-IV & V). SCT technology allows transfection complex easier and more efficient in crossing and penetrating the cell membrane, leading to 3~15 times better efficiency than that on unshaved cells (Figure 3-V & VI). The SCT technology is extremely useful for transfecting the hard-to-transfection cells.
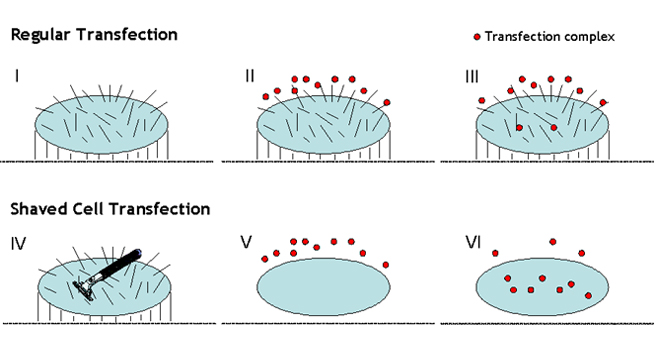
Figure 3. A Cartoon Showing How Shaved Cell Transfection Technology Works
4. Transfection Mediated Cytotoxicity Removal (TMCR) Technology:
Researches often encounter the following dilemma during DNA/siRNA transfection operations: transfection reagent with high efficiency usually gives strong cytotoxicity. Sometimes the toxicity is so strong that large amount of cells are killed after transfection. Therefore researchers have to sacrifice transfection efficiency to trade for relative healthy cells. Now we provide a solution to achieve maximum transfection efficiency while keeping relative good cell viability by removing transfection mediated cytotoxicity which otherwise kills the cells. After years of research, we identified that it is the transfection complex sticking to the edges that leads to cytotoxicity (Figure 4, upper panel). Due to the nature of transfection complex, it is impossible to wash the complex out with just medium change post transfection. We develop an unique product in which several chemicals are mixed to make a cocktail (Patent pending, US PTO # 62375686) which was confirmed to effectively remove all the transfection complex bound to cell edges with 5 minutes incubation at room temperature (Figure 4, lower panel), thus removing the transfection mediated cytotoxicity.
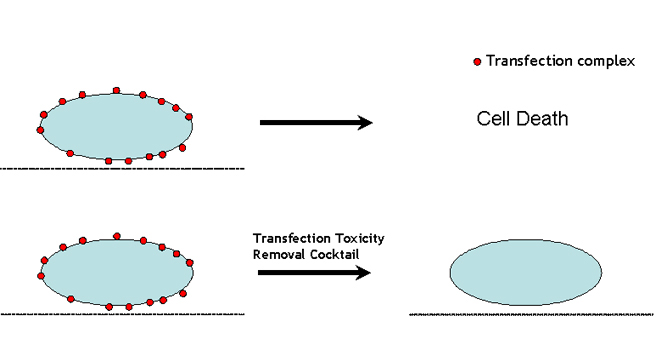
Figure 4. A Cartoon Showing How Transfection Mediated Cytotoxcity Removal (TMCR) Technology Works
5. pH Dependent Conformational Change (PDCC) Technology for Efficient siRNA Delivery:
While liposome or polymer reagents often give very good DNA delivery efficacy, they failed to efficiently drive siRNA into mammalian cells. The poor performance of liposome or polymer based siRNA transfection reagents could be linked to the length of the siRNA anionic segment that is too short to maintain electrostatic cohesion with the cationic lipids or polycationic polymer. Consequently, siRNA lipoplex or polyplex breaks apart too easily upon touching polyanionic cell surface. We modified the liposome and polymer with specific hydrophobic groups which confers the pH dependent conformational changes (PDCC) at physiological pH condition and greatly stabilizes siRNA lipolplex or polyplex. The equal important property resulting from PDCC technology is that siRNA lipoplex or polyplex nanoparticles can be controlled to be virus-like size by adding specific hydrophobic groups, leading to much better siRNA transfection efficiency.
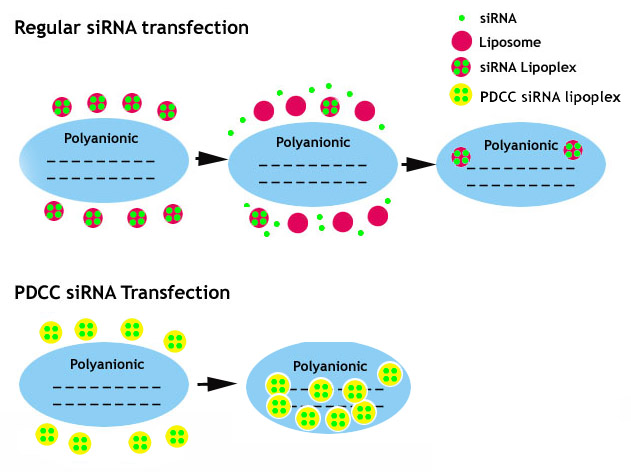
Figure 5. A Cartoon Showing How pH Dependent Conformational Change (PDCC) Technology Works
6. Ad.MAX™ Technology for Maximum Adenovirus Production:
Ad.MAX™ technology was developed by genetically modifying virus packaging cell, HEK293 cell and adenoviral shuttle vector or adenoviral genome for maximum adenovirus production. The core of this proprietary technology lies in the genetically engineered HEK293 in which adenoviral replication is kept intact while viral protein expression during adenovirus packaging process is significantly suppressed. In combination with a trans-element in adenovirus genome which recognizes the suppressor cassette of HEK293 cell, Ad.MAX™ system allows maximum adenovirus production with minimum protein expression during viral packaging. This system is extremely useful for packaging adenoviral vectors with toxic genes of interests (GOI), which will otherwise kill HEK293 during adenovirus replication and production (Figure 6), leading to dramatic reduction in adenovirus yield. Ad.MAX™ technology eventually allows researchers to construct all genes (<7.5 kb) to adenovirus.
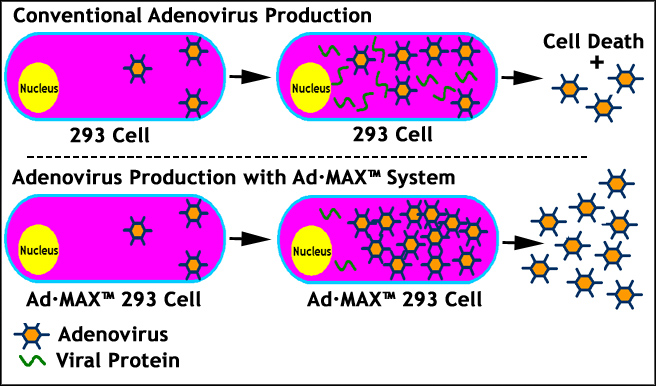
Figure 6. A Cartoon Showing How Ad.MAX™ Technology Works
7. Multi Approaches to Produce Super Infectious rAAV Particles at Super High Titer:
Not like adenovirus, it is relatively harder to achieve high yield of rAAV with regular rAAV production procedures. By adopting the following multi strategies, we have managed to produce super infectious (~30 times better infectious capability than conventional rAAV) rAAV particles at super high titer (up to 1E+15 GC).
– AAV·HT™ Packaging Cell: We have screened different HEK293 cell types and developed a highly productive strain——AAV·HT™ packaging cell which per our validation data produces ~10 times more rAAV particles than regular HEK293.
– Modify rAAV cis Vector: Customer has an option to choose rAAV cis vector with a truncated WPRE cassette downstream of transgene. Inclusion of WPRE cassette in rAAV cis vector allows to produce ~8 times more rAAV particles than rAAV cis vector without WPRE.
– Modify rAAV Capsid: rAAV capsid mutations at several specific sites significantly (~30 folds better in vitro & in vivo) enhanced rAAV transduction efficiency. Customers have an option to choose packaging rAAV with mutant capsids to produce more infectious rAAV vector.
– Package Double-stranded AAV (dsAAV): Double-stranded AAV (dsAAV) also known as self-complementary AAV (scAAV) is confirmed to deliver up to 50 times better transduction efficiency in both vitro and vivo. Custom service is available to generate and package gene of interests to a scAAV vector*.
– Advanced Double CsCl Ultra-centrifugation Protocol: The main feature of the advanced protocol is treatment with a special formulated detergent after 3xfreeze/thaw cycle and pre-precipitation step before loading to ultra-centrifugation. The advanced protocol produces super purified (clinical trial grade) and super infectious rAAV vector at high yield. See Figure 8 for details. The differences with conventional protocol are being framed in red.
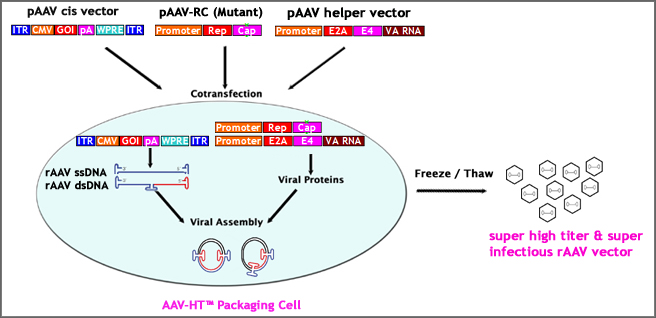
Figure 7. Multi Approaches to Produce Super Infectious rAAV Particles at Super High Titer
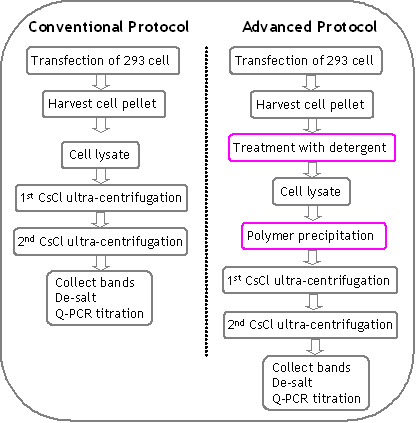
Figure 8. Outline and comparison of advanced protocol and conventional protocol. The differences in advanced protocol are being framed in red.
